- Blog
- About
- Contact
- Rooney new sinhala song
- Canon lbp 2900 printer software free download for windows 8
- Hummer h3 interiors
- Corel videostudio ultimate x10 better than adobe premiere
- Semisonic closing time live
- Eyeframe converter dnxhd no proxy
- Ragdoll masters unblocked
- Gangs of wasseypur putlockers
- Wic reset utility 2
- Xbox sega games
- Word game popcap games
- Anker docking station driver for mac
- Kumbalangi nights reviews
- Mega man x4 boss weaknesses
- Acetaldehyde ternary diagram
- Portrait professional mac torrent
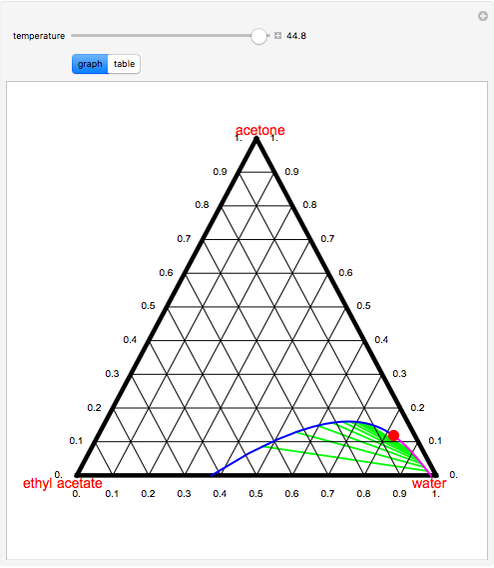
When nanoscale ZnO is present, the resulting composite shows preferential hydrogen and CO 2 formation due to the fact that the dehydrogenation and total oxidation pathway is more favoured compared to the binary composite. A new anhydrous phase in the system is described, which was previously thought to be synthetic andalusite it is probably a new polymorph of the Al 2 SiO 5 composition with ortho-rhombic unit-cell dimensions a7.55 A, b8.27 A, and c 5.66 A. While using binary (13.8 wt% Cu) as catalyst, acetaldehyde was obtained with a yield of 87% at 355 ☌ (selectivity 96% and conversion 91%). Revised Phase Diagram for the System Al2O3SiO2 - ARAMAKI. The catalytic activities of the and binary and ternary composites were studied for the selective partial oxidation of ethanol to acetaldehyde with molecular oxygen. SEM and TEM of ternary clearly reveal that Cu 2O nanoparticles are primarily deposited on ZnO rather than on MWCNTs. Transmission electron microscopy ( TEM) of the sample shows crystalline domains of size 4–10 nm surrounded by an amorphous region within the larger particles. Liquid-liquid equilibrium data for the ternary systems of toluene+water+propionic acid, o-xylene+water +propionic acid, methyl isobutyl ketone+water+propionic acid, ethyl acetate+water+propionic acid, and 1-butanol +water+propionic acid were measured at 25 C and atmospheric pressure. Then the corresponding green tie line is drawn. Click one of the two curves to place the locator. Ternary phase equlibirum data for glycerol-water-alcohol systems and extraction of glycerol from spent soap lye using alcohols as. The composition of the heavy and light phases are shown in blue and red respectively. Ternary phase equilibrium data for tetrahydrofuran-water-solvent systems and separation of tetrahydro furan from aqueous solution. Scanning electron microscopy ( SEM) of the binary nano-composite shows an increase of cuprous oxide loading depending on the precursor amount, along the periphery of the MWCNTs as well as formation of larger particle agglomerates. This Demonstration displays the liquid-liquid equilibrium diagram for a chloroform-water-acetic-acid mixture at 25☌ and 1 atm. Thermogravimetric coupled mass spectroscopic analysis ( TG-MS) of 1 revealed that methanol formed during the decomposition of 1 acts as a potential in situ reducing agent. The precursor decomposes either in air or under argon to yield CuO 2 by in situredox reaction.

Di-aqua-biscopper Cu 2♲H 2O 1 in DMF was used as single source precursor for the deposition of nanoscaled Cu 2O. Cuprous oxide agglomerates composed of 4–10 nm Cu 2O nanoparticles were deposited on multiwalled carbon nanotubes (MWCNTs) and on ZnO/MWCNTs to give binary and ternary composites.
